Saudi Arabia Medical Device Approval | SFDA Regulations

Medical Device & IVD Registration in Saudi Arabia: A Complete Guide
The Saudi Food & Drug Authority (SFDA) is the national regulator responsible for medical devices in KSA. SFDA issues the Medical Device Market Authorization (MDMA) and maintains requirements, guidance, and online services for device registration and establishment licensing. All device registration and related administrative activities are managed through SFDA e-services (MDMA / GHAD systems).
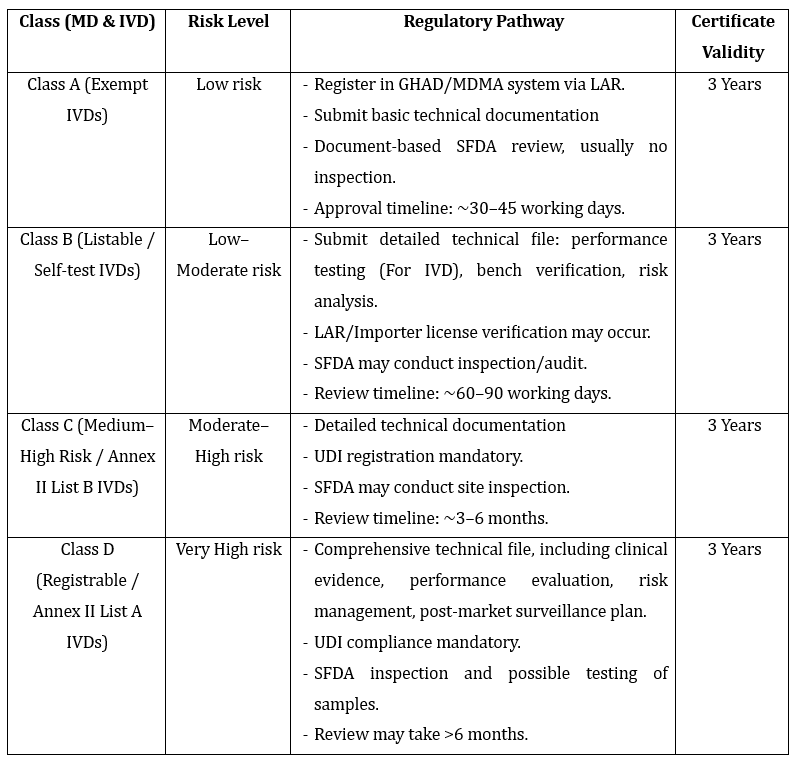
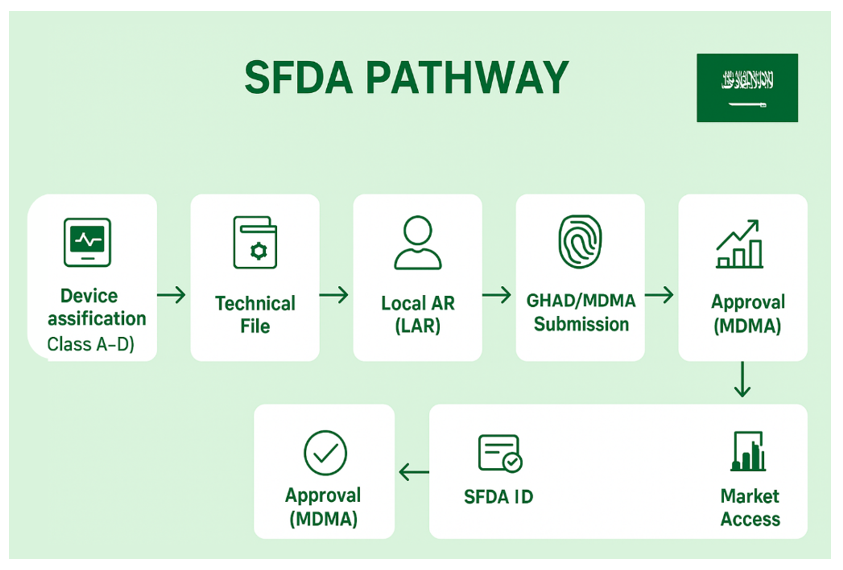
1. SFDA Device Classification & Registration Pathway
2. Appoint a Local Authorized Representative (LAR):
- Foreign manufacturers must designate a Saudi-based LAR to act as the official liaison with the SFDA.
- The LAR:
- Acts as the legal contact point with SFDA,
- Submits applications on your behalf,
- Receives regulatory communications, and
- May need a local license to act as AR (SFDA requires AR registration/licensing).
- Ensure compliance with SFDA regulations
3. Prepare Technical Documentation:
- Compile a comprehensive technical file, including:
- Device description and specifications
- Instructions for Use (IFU)
- Design and manufacturing information
- Safety and performance evidence
- Product testing and clinical data
- Risk management file
- Labelling and post-market information
- Higher-risk devices may require additional documentation, such as clinical data and performance testing results.
4. QMS requirements by device class
The QMS requirements vary depending on the device's risk classification:
- For all device classes: Manufacturers, importers, and distributors must establish and maintain an effective QMS.
- For Class A (non-sterile/non-measuring) and Class I (low-risk) devices: A QMS certificate from a Saudi Conformity Assessment Body (CAB) is typically required.
- For Class C and D (high-risk) devices: The SFDA may require an inspection or audit of the manufacturer's QMS in addition to the ISO 13485 certificate
5. Submit Application via GHAD System:
SFDA uses an e-service (MDMA / GHAD) for device and manufacturer registration and for MDMA submission. Manufacturer or the AR must create accounts and register manufacturer and devices in the system prior to submitting the MDMA. Typical submission steps:
- Register manufacturer & manufacturing site in MDMA/GHAD.
- Register the device record (device master).
- Upload the technical dossier and required certificates.
- Pay the application fees (fee schedule depends on class and number of products).
- Submit and track the application in the portal.
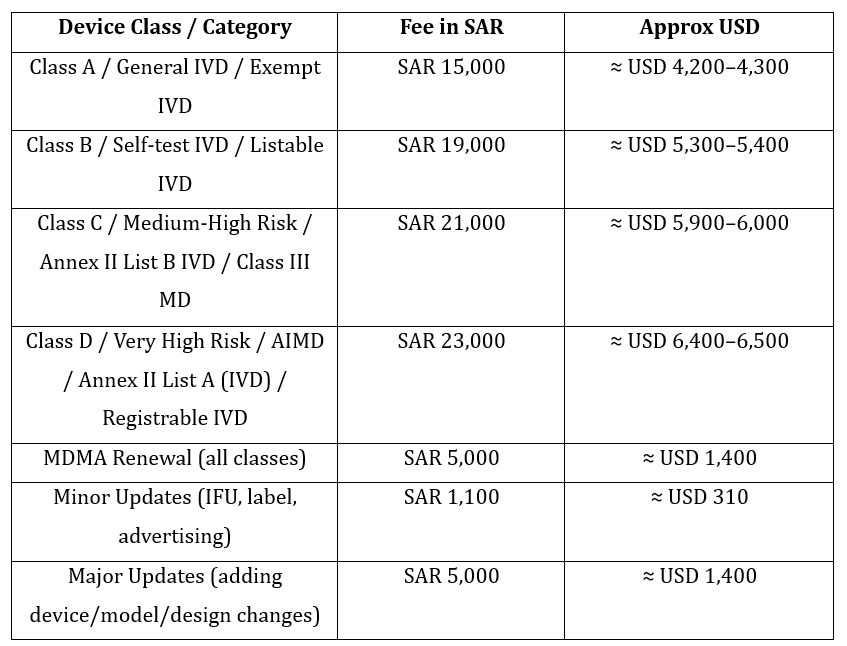
6. Fee requirements:
7. Inspections / audits
SFDA may perform site inspections for higher-risk devices (Class B/C/D), manufacturing facilities, or establishment licensing. Inspections evaluate compliance with Good Manufacturing/Storage Practices and the claims in your dossier.
8. Obtain approval & market the device
- Respond promptly to SFDA deficiency letters/questions in the MDMA portal.
- If SFDA requires an inspection, coordinate with your AR and manufacturing site to host SFDA inspectors or provide requested evidence.
- After SFDA grants the MDMA, you receive authorization to market the device in KSA.
- Ensure your establishment license (importer/distributor/warehouse) is active for entities that will hold and sell the device in KSA.
9. Facility (establishment) registration
Manufacturing sites, importers, distributors, warehouses, and establishments that handle medical devices must be licensed/registered with SFDA. Establishments may need to meet premises, storage, and record requirements and upload supporting documents (title deed/lease, site layout, responsible person details).
10. UDI (Unique Device Identification) requirements
SFDA has implemented a UDI system and requires manufacturers to comply with SFDA UDI requirements. The SFDA UDI system (Saudi-DI) defines UDI rules, issuer standards, data elements, and the national UDI database submission requirements. UDI obligations include placing UDI on device labels and submitting UDI data to the SFDA database according to the timelines and class-based phases.
11. References
Medical Device Facility Licensing: Medical Device Facility Licensing | The official website of the Saudi Food and Drug Authority
Share this blog
Read More Blogs

Medical Device Registration in Brazil | ANVISA Process

Medical Device Approval Process in Japan | PMDA and MHLW Guide


Is DHF Compliance Risks Holding Back Your Medical Device Approval?