Medical Device Registration in Brazil | ANVISA Process

1. Medical Device Approval in Brazil: Complete ANVISA Guide
In Brazil, the Brazilian Health Regulatory Agency (ANVISA) plays a central role in ensuring the safety and efficacy of medical devices. ANVISA categorizes Medical Devices into four types: medical equipment, materials for health use, orthopaedic implants and in vitro diagnostic medical devices.
2. Device Classification
The medical devices, object of this Resolution, are classified according to the intrinsic risk they represent to the health of the user, patient, operator, or third parties involved, in Classes I, II, III, or IV:
- ✔️
I - Class I: low risk; subject to notification
- ✔️
II - Class II: medium risk; subject to notification
- ✔️
III - Class III: high risk; subject to marketing authorization.
- ✔️
IV - Class IV: maximum risk; subject to marketing authorization.
These classifications are determined based on the device's intended use and associated risks, following the 22 classification rules outlined in Annex I of the resolution. Devices and their accessories must be classified separately. If a device is intended for use in combination with another, each component is assessed individually to determine its appropriate classification. ANVISA categorizes Medical Devices into four types: medical equipment, materials for health use, orthopaedic implants and in vitro diagnostic medical devices.
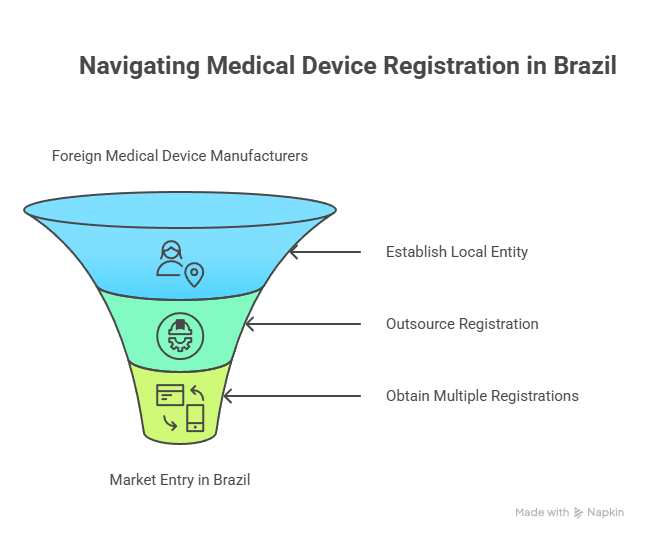
3. Brazil Market Access: Local Representation for Foreign Manufacturers
Only Brazilian-registered companies are eligible to apply to ANVISA for licenses to manufacture or distribute medical devices. Foreign manufacturers cannot apply directly and must appoint a licensed local importer to submit their notifications or registrations on their behalf. Since there's no “Authorized Rep” role like in Europe, you've got three winning moves:
- Set up your own licensed entity with B-GMP compliance.
- Partner with a Brazil Registration Holder (BRH) for flexible, controlled access.
- Register through multiple importers—best for low-risk devices.
4. Device Registration in Brazil: ANVISA
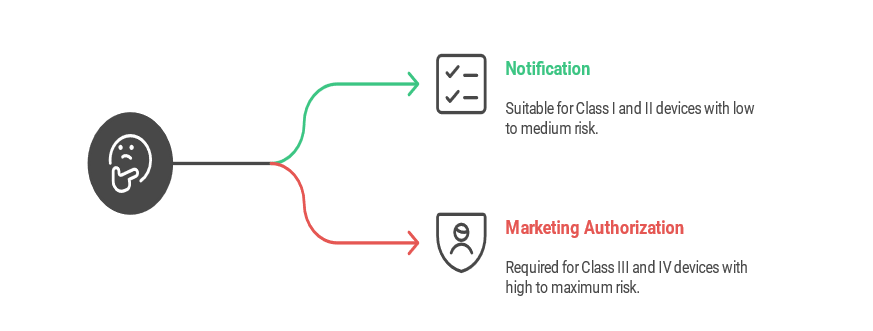
In Brazil, medical device approval follows two main pathways based on device classification: Notification for lower-risk devices and Marketing Authorization for higher-risk devices. The chosen pathway determines the regulatory requirements, documentation, and review process required by ANVISA.
4.1 Application for notification
In Brazil, Class I and II medical devices follow the streamlined notification pathway. This simplified process requires a product-specific form, a Letter of Authorization from the foreign manufacturer to the Brazil Registration Holder (BRH), and any relevant Brazilian certifications. While a Technical Dossier—covering risk management, global marketing history, usability studies, and compliance—is required, it stays on file and isn't submitted with the application. Notifications remain valid indefinitely, though any device changes must be reported to ANVISA before implementation.
4.2 Application for Marketing Authorization
The Marketing Authorization pathway in Brazil, applies to Class III and IV medical devices. This route requires a comprehensive submission of technical and clinical data to demonstrate the device's safety and efficacy, sometimes including pivotal clinical study results for high-risk products like drug-eluting stents. Compliance with Brazilian Good Manufacturing Practices (B-GMP) is mandatory before approval, though applications can be submitted beforehand. Once approved, registrations are valid for ten years, and renewal applications must be submitted at least 180 days prior to expiration. This process ensures rigorous oversight for high-risk devices entering the Brazilian market.
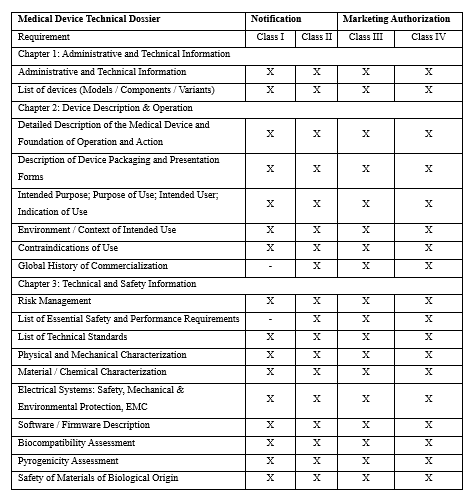
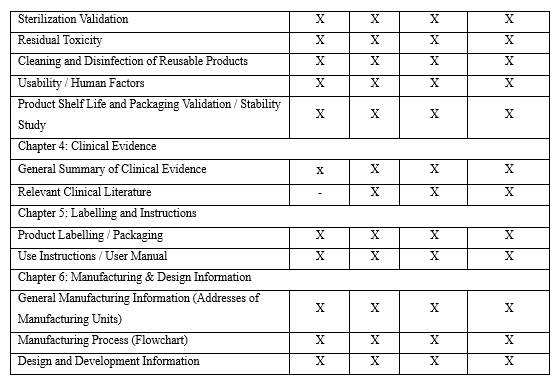
5. Technical Dossier Structure of Medical Devices: ANVISA
6. Cost and time
ANVISA fees for Medical Devices:
- Class I and II (Low risk): About USD 190,00
- Class III and IV (High Risk): About USD 890,00
- GMP Certification: About USD 25.890,00
Time of ANVISA review (After submission) and testing:
- Class I: 15 days
- Class II: About 2 months
- Class III and IV: 4 - 5 months
- GMP Certification: About 8 months.
7. References
- ANVISA. RDC 751/2022 - Medical devices: Materials for health use and Orthopedic Implants.
- ANVISA. RDC 830/2023 - In vitro diagnostics.
- ANVISA. RDC 591/2021 - Unique Device Identification (UDI).
- ANVISA. RDC 657/2022 - Software as a Medical Device (SaMD).
- Fees & Timeline: Medical device registration in Brazil. ANIVISA | Inmetro | Anatel
Share this blog
Read More Blogs

US FDA Medical Device Registration Guide | 510(k), PMA & De Novo


Gap Assessment: ISO 11137-1:2025 vs ISO 11137-1:2006


The Medical Device Single Audit Program