CDSCO Medical Device Registration in India – CDSCO SUGAM Portal Guide

Step-by-Step Guide to Register a Medical Device in India
The Indian medical device market is rapidly expanding, driven by advancements in technology and increasing healthcare demand. If you are a manufacturer or distributor planning to enter India, understanding the regulatory requirements is crucial. The Central Drugs Standard Control Organization (CDSCO) is the primary regulatory authority responsible for medical device registration in India. Here’s a detailed guide to help you navigate the registration process.
1. Regulatory Body: CDSCO
All medical device registrations in India are managed by the CDSCO, which has a dedicated online portal called SUGAM for submitting applications. The portal facilitates online submission for import licenses, manufacturing licenses, and registration certificates.
The CDSCO operates under the Ministry of Health and Family Welfare and coordinates with both Central Licensing Authority (CLA) and State Licensing Authorities (SLA) depending on the type of device.
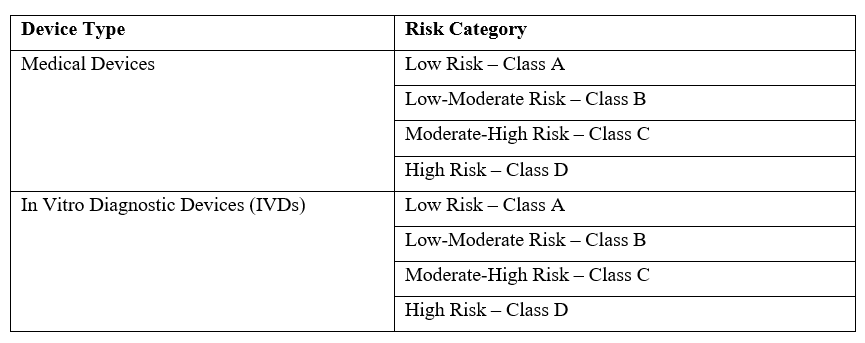
2. Device Classification
India follows a risk-based classification system for medical devices, as outlined in the Medical Devices Rules, 2017 (MDR 2017): The classification determines the regulatory pathway, licensing authority, and documentation required for registration.
3. Regulatory Pathway
The registration process in India typically involves the following steps:
Step 1: Appoint an Indian Authorized Representative (AR)
Foreign manufacturers must appoint an Indian Authorized Agent, who acts as the primary liaison with CDSCO. The AR is responsible for all regulatory communication and application submissions in India.
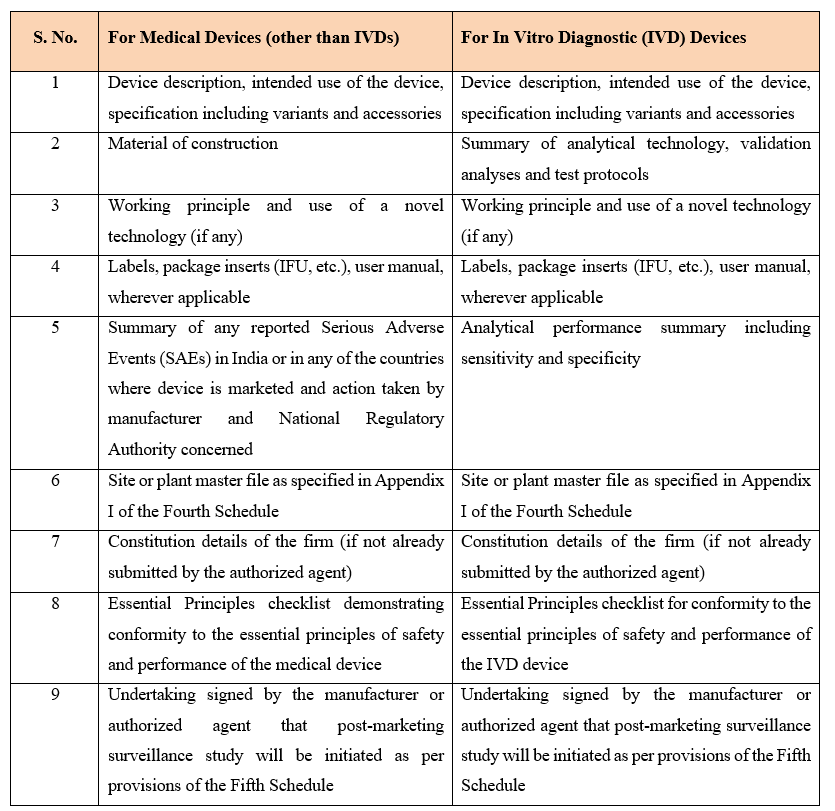
Step 2: Prepare a Technical Dossier
The technical dossier provides detailed information about the device, including:
Step 3: Implement a Quality Management System (QMS)
Manufacturers are required to maintain a Quality Management System that complies with regulatory standards. A robust QMS ensures that the device is consistently produced and controlled according to quality standards.
Step 4: Obtain Free Sale Certificate (For Import Applications)
Foreign manufacturers must submit a Free Sale Certificate or equivalent regulatory approval from their country of origin. This demonstrates that the device is legally marketed in the home country.
Step 5: Register on the SUGAM Portal
All applications for device registration, import, and manufacturing licenses must be submitted online through the SUGAM portal. Steps include:
- Create a user account on SUGAM
- Upload the application, technical dossier, and supporting documents
Step 6: Submit the Application
- Submit the completed application for the appropriate license
Step 7: CDSCO Review and Approval
The timeline for regulatory review depends on the device class
Step 8: Inspection (If required)
- Class A: Inspection may occur within 120 days after license issuance
- Class B: Audit of manufacturing site by Notified Body within 90 days
- Class C & D: Inspection by CLA within 60 days
Conclusion
Registering a medical device in India is a structured process involving classification, technical documentation, quality management, online submission via SUGAM, and approval by CDSCO. Understanding each step is essential to ensure compliance, reduce delays, and successfully bring your device to the Indian market.
With proper planning, appointment of an Indian Authorized Representative, and adherence to the MDR 2017, manufacturers can navigate the regulatory pathway efficiently and gain timely approval for marketing or importing medical devices in India.
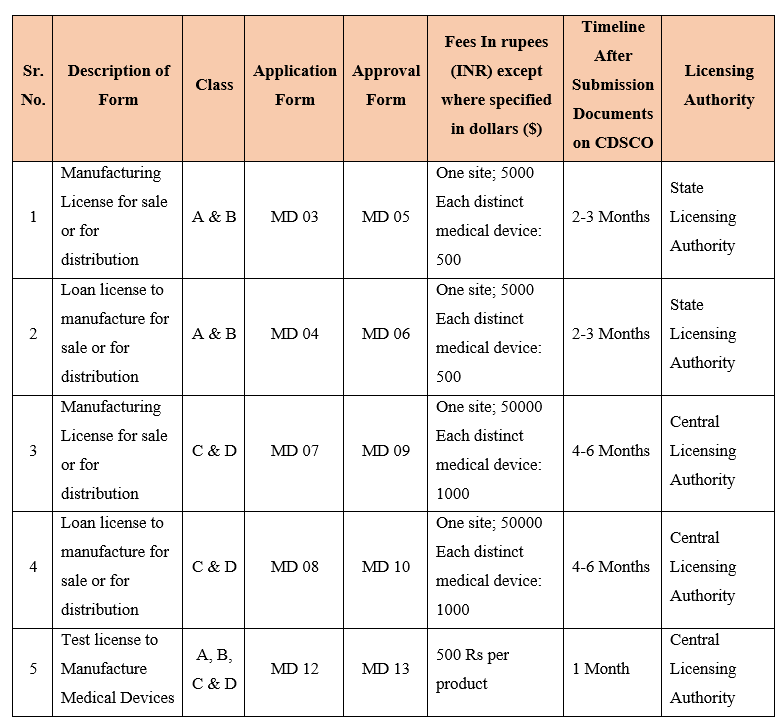
CDSCO Forms Under MDR 2017
| Form No | Description |
|---|---|
| Form MD1 | Application for grant of certificate of registration of a notified body |
| Form MD3 | Application for grant of license to manufacture for sale or for distribution for Class A or Class B medical device |
| Form MD4 | Application for grant of loan license to manufacture for sale or for distribution of Class A or Class B medical device |
| Form MD5 | Licence to manufacture for sale or for distribution of Class A or Class B Medical Device |
| Form MD6 | Loan Licence to manufacture for sale or for distribution of Class A or Class B Medical Device |
| Form MD7 | Application for grant of license to manufacture for sale or for distribution of Class C or Class D medical devices |
| Form MD8 | Application for grant of loan license to manufacture for sale or for distribution of Class C or Class D medical device |
| Form MD9 | Licence to manufacture for sale or for distribution of Class C or Class D Medical Device |
| Form MD10 | Loan Licence to manufacture for sale or for distribution of Class C or Class D Medical Device |
| Form MD11 | Form in which the audit or inspection book shall be maintained |
| Form MD12 | Application for license to manufacture medical device for purpose of clinical investigations, test, evaluation, examination, demonstration, or training |
| Form MD13 | Licence to manufacture Medical Device for the purpose of clinical investigations or test or evaluation or demonstration or training |
| Form MD14 | Application for issue of import license to import medical device |
| Form MD15 | Licence to import Medical Device |
| Form MD16 | Application for license to import medical devices for the purposes of clinical investigations or test or evaluation or demonstration or training |
| Form MD17 | Licence to import medical devices for the purposes of clinical investigations or test or evaluation or demonstration or training |
| Form MD18 | Application for license to import investigational medical devices for the purposes by a government hospital or statutory medical institution for the treatment of patients |
| Form MD19 | Licence to import investigational medical devices for the purposes by a government hospital or statutory medical institution for the treatment of patients |
| Form MD20 | Application for permission to import small quantity of medical devices for personal use |
| Form MD21 | Permission to import small quantity of medical devices for personal use |
| Form MD22 | Application for grant of permission to conduct clinical investigation of an Investigational Medical Device |
| Form MD23 | Permission to conduct clinical investigation of an Investigational Medical Device |
| Form MD24 | Application for grant of permission to conduct clinical performance evaluation of New In-Vitro Diagnostic Medical Device |
| Form MD25 | Permission to conduct clinical performance evaluation of New In-Vitro Diagnostic Medical Device |
| Form MD26 | Application for grant of permission to import/manufacture for sale or for distribution of medical device which does not have a predicate medical device |
| Form MD27 | Permission to import/manufacture for sale or for distribution of medical device which does not have a predicate medical device |
| Form MD28 | Application for grant of permission to import or manufacture for sale or for distribution of a New In-Vitro Diagnostic Medical Device |
| Form MD29 | Permission to import or manufacture New In-Vitro Diagnostic Medical Device |
| Form MD30 | Memorandum to Central Medical Device Testing Laboratory |
Share this blog
Read More Blogs

The Medical Device Single Audit Program


Canada Medical Device Regulatory Pathway


Medical Device Registration in Brazil | ANVISA Process