TGA Medical Device Registration: Step-by-Step Guide for Australia

How to Register Medical Devices and IVDs in Australia: A Complete Guide
Australia has a robust regulatory system to ensure the safety, quality, and performance of medical devices and in-vitro diagnostic devices (IVDs). The Therapeutic Goods Administration (TGA) is the Australian government authority responsible for evaluating, assessing, and monitoring therapeutic goods, including medicines, medical devices, and biologicals. Compliance with TGA regulations is mandatory to legally supply medical devices in Australia.
This guide provides a step-by-step overview of the regulatory process for registering medical devices and IVDs in Australia.
1. Understanding the Regulatory Body: TGA
The TGA regulates medical devices and IVDs to protect public health and safety. Its responsibilities include:
- Assessing the safety and performance of devices.
- Monitoring devices after they are on the market.
- Maintaining the Australian Register of Therapeutic Goods (ARTG), a public database of legally supplied therapeutic goods.
For more details: TGA Official Website
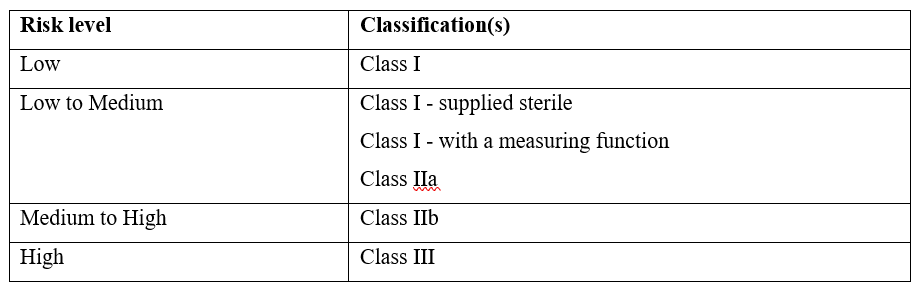
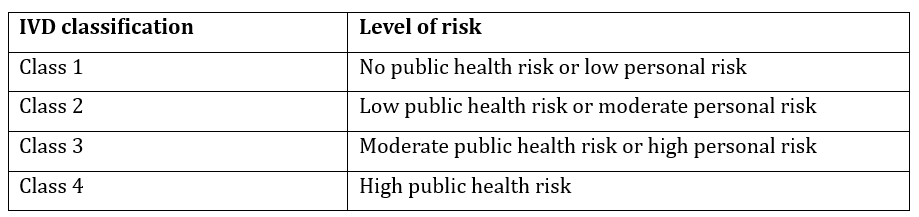
2. Device Classification
Medical devices and IVDs are classified based on the risk they pose to patients. Device classification determines the regulatory requirements, including conformity assessment and documentation
- Medical Devices (MDs): Refer to TGA Medical Device Classification
- In-Vitro Diagnostic Devices (IVDs): Refer to TGA IVD Classification
3. Appoint an Australian Sponsor
To register a device in Australia, a sponsor must be appointed.
- The sponsor is responsible for submitting applications to the TGA and ensuring compliance with regulatory requirements.
- A sponsor must be:
- A resident of Australia, or
- An incorporated body in Australia conducting business in the country with a representative residing locally.
For more information: Role of the Sponsor
4. Demonstrate Conformity to Essential Principles
Manufacturers must demonstrate that their devices comply with the Essential Principles of safety and performance, which include:
- Ensuring devices are safe for intended use.
- Generating and maintaining scientific and clinical evidence.
- Performing risk management and design verification.
Checklist reference: TGA Essential Principles
5. Steps for Device Sponsors and Manufacturers
Sponsors:
- Prepare documentation demonstrating conformity to Essential Principles.
- Submit applications for device inclusion in the ARTG.
- Ensure post-market obligations are met.
Manufacturers:
- Perform conformity assessment procedures.
- Maintain quality management systems.
- Provide evidence of device safety and performance.
Steps for sponsors: TGA Steps for Device Sponsors Steps for manufacturers: TGA Steps for Device Manufacturers
6. Prepare Documentation
Required documentation typically includes:
- Device description and classification
- Conformity assessment certificates
- Clinical evidence supporting safety and performance
- Labelling and instructions for use
- Risk management files
- Post-market surveillance plans
7. Priority Review Pathway
The TGA provides a priority review pathway for medical devices, which accelerates assessment for high-priority applications. This pathway applies to:
- Conformity assessment certificate applications
- ARTG inclusion applications
Priority applications receive front-of-queue consideration throughout the assessment process.
More info: Priority Review Pathway
8. Submit the Application
Applications are submitted via the TGA Business Services (TBS) portal, which allows:
- Managing registration applications
- Viewing, cancelling, or updating entries
- Downloading certificates and ARTG entries
Portal link: TGA Business Services
9. Application Review and ARTG Inclusion
- All devices must be included in the ARTG before being lawfully supplied in Australia.
- ARTG is a public database listing all therapeutic goods approved for supply.
For more info: ARTG Overview
10. Unique Device Identification (UDI)
Australia is implementing UDI to strengthen patient safety. UDI enables:
- Identification of medical devices
- Improved post-market monitoring
- Efficient management of recalls
For details: TGA UDI Hub
11. Labelling Requirements
Medical devices must comply with Essential Principle 13, which mandates:
- Accurate information on device use
- Clear instructions for use
- Safety and performance information
Regulatory reference: Therapeutic Goods (Medical Devices) Regulations 2002
12. Post-Market Obligations
Post-market monitoring ensures devices remain safe in real-world use. Key responsibilities include:
- Investigating adverse events
- Implementing corrective and preventive actions
- Reporting incidents to the TGA
- Maintaining risk management procedures
Guidance: Post-Market Responsibilities
13. Conclusion
Registering a medical device or IVD in Australia involves a systematic approach:
- Determine classification and appoint an Australian sponsor.
- Demonstrate conformity with Essential Principles.
- Prepare documentation and submit via the TGA Business Services portal.
- Include the device in the ARTG.
- Comply with UDI, labelling, and post-market requirements.
Following these steps ensures compliance with the TGA and enables the legal supply of medical devices and IVDs in Australia.
Share this blog
Read More Blogs

Understanding ICMED: India's Certification for Medical Devices


FDA QMSR Explained: Key Changes & ISO 13485 Alignment (2026)


EU MDR and IVDR Medical Device Regulatory Pathway