SAHPRA Medical Device Registration: Step-by-Step Guide

A Complete Guide to Registering a Medical Device in South Africa
South Africa is one of the fastest-growing healthcare markets in Africa, making it an attractive for medical device manufacturers and suppliers. However, before entering this market, medical device and IVD (In Vitro Diagnostic) manufacturers must comply with the regulations set forth by the South African Health Products Regulatory Authority (SAHPRA).
This blog will walk you through the step-by-step process of registering a medical device in South Africa, including classification, licensing, documentation, and vigilance requirements.
1. Regulatory Authority in South Africa
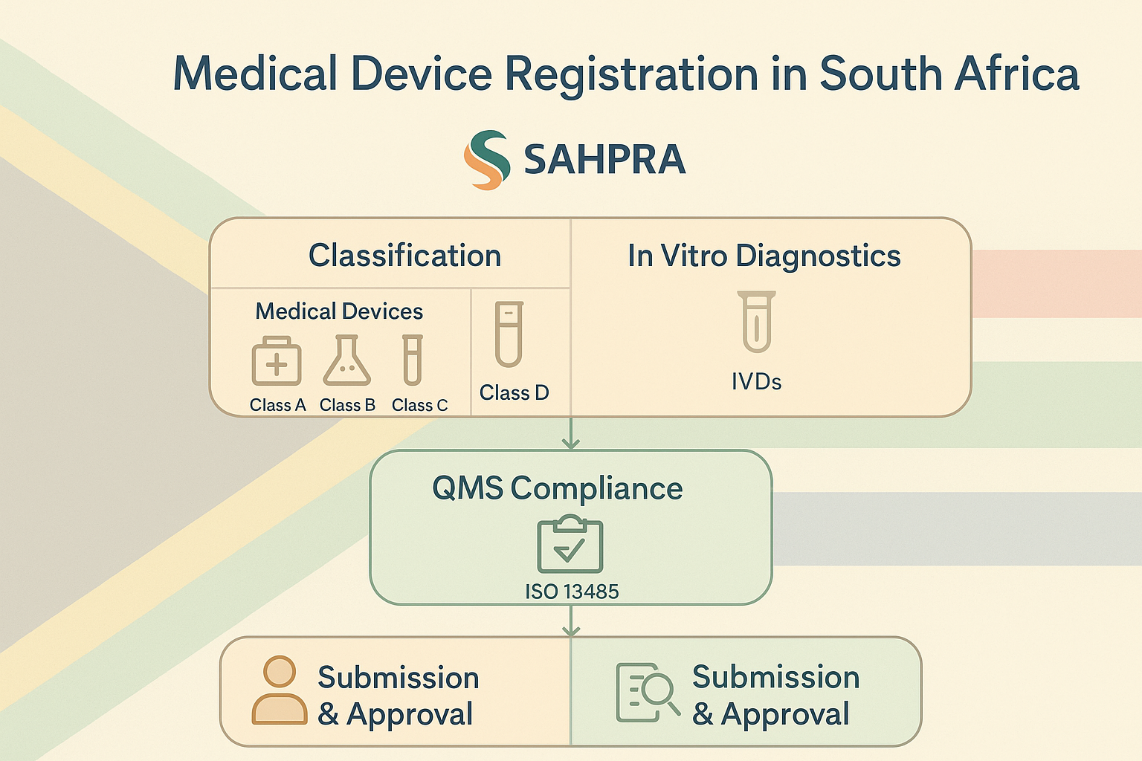
The South African Health Products Regulatory Authority (SAHPRA) is the official body responsible for regulating medical devices and IVDs. It ensures that only safe, effective, and high-quality products reach the market.
2. Device Classification
Medical devices and IVDs are classified based on risk, aligning with international practices:
Medical Devices
- Class A: Low risk
- Class B: Low-moderate risk
- Class C: Moderate-high risk
- Class D: High risk (impact on patients or public health)
In Vitro Diagnostics (IVDs)
- Class A: No public health risk or low personal risk
- Class B: Low public health risk or moderate personal risk
- Class C: Moderate public health risk or high personal risk
- Class D: High public health risk
3. Registration Pathway
Currently, the registration process for medical devices is still under development. SAHPRA has introduced a Registration Call-Up Plan, which specifies when different device classes will be called up for registration.
SAHPRA also follows reliance pathways, where approval in recognized jurisdictions such as the EU, USA, Canada, Brazil, Japan, Australia, or WHO Prequalification for IVDs can support faster registration in South Africa.
4. Quality Management System (QMS)
Manufacturers must demonstrate compliance with internationally accepted quality standards.
- ISO 13485 certification is required.
- A Quality Manual must be submitted with the application.
5. Obtain an Establishment Licence
Before registering products, companies must apply for the appropriate medical device establishment licence:
- GLF-MD-06A: Manufacture medical devices
- GLF-MD-06B: Wholesale medical devices
- GLF-MD-06C: Import, distribute, or export medical devices
Licensing applies to Class B, C, and D devices. The process also requires submission of a Quality Manual or Site Master File, depending on the applicant's role.
6. Appoint a South African Authorized Representative (AR)
Foreign manufacturers must appoint a local Authorized Representative (AR) in South Africa. The AR is responsible for liaising with SAHPRA and ensuring regulatory compliance.
7. Required Documentation
Business as Usual (BAU) Applications
- Cover Letter
- Licence Application Form (Annex 1)
- Proof of Payment (Annex 2)
- CV of the Authorized Representative (Annex 3)
- Quality Manual (Manufacturers/Distributors) or Site Master File (Wholesalers)
8. Post-Market Surveillance & Vigilance
Manufacturers and distributors must report medical device adverse events within strict timelines:
- 48 hours: Serious public health threat
- 10 days: Death or serious health deterioration
- 30 days: Events that could potentially cause death or serious deterioration if repeated
9. Making Changes to Applications
- Notification: For minor updates (e.g., GMDN codes, manufacturer details, adding products of the same class). No fees currently apply.
- Amendment: For major changes that affect licence details. Applicable fees apply (as published by SAHPRA).
10. Conclusion
South Africa offers significant opportunities for medical device companies, but regulatory compliance is essential for market entry. With SAHPRA strengthening its framework and aligning with international best practices, early preparation and a clear understanding of the pathway will help ensure a smoother registration process. If you're planning to register your device in South Africa, now is the time to prepare your documentation, QMS certification, and establish local representation.
11. Reference
- SAHPRA Medical Devices Portal
- SAHPRA Guideline for Classification of Medical Devices & IVDs
- Guideline on Medical Device Quality Manual
- Application Forms - SAHPRA
- Medical Device Adverse Event Reporting Form
- Guideline for Adverse Event Reporting
Share this blog
Read More Blogs

EU MDR and IVDR Medical Device Regulatory Pathway


Canada Medical Device Regulatory Pathway


Understanding ICMED: India's Certification for Medical Devices